Cytologic Screening: Detecting Cellular Changes Before Symptoms Appear
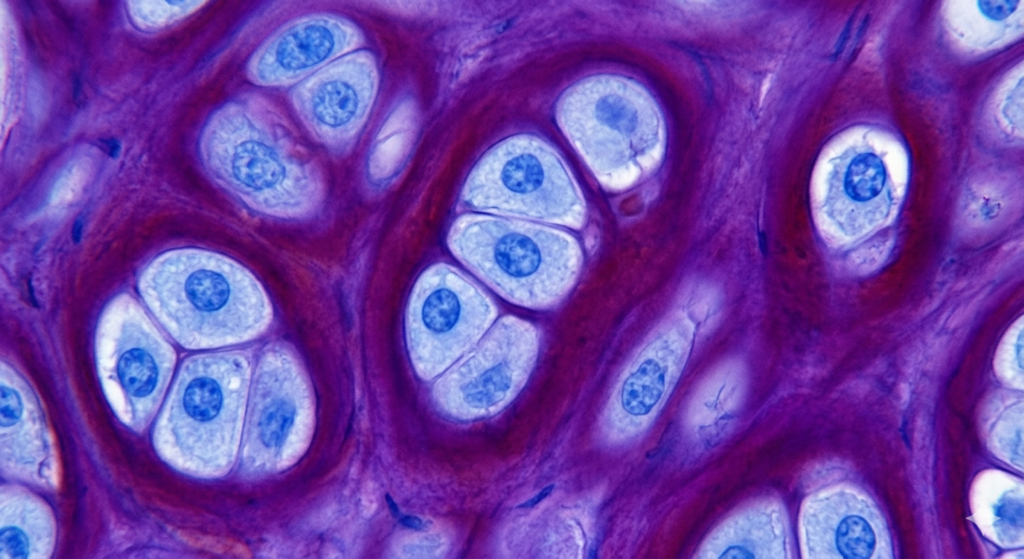
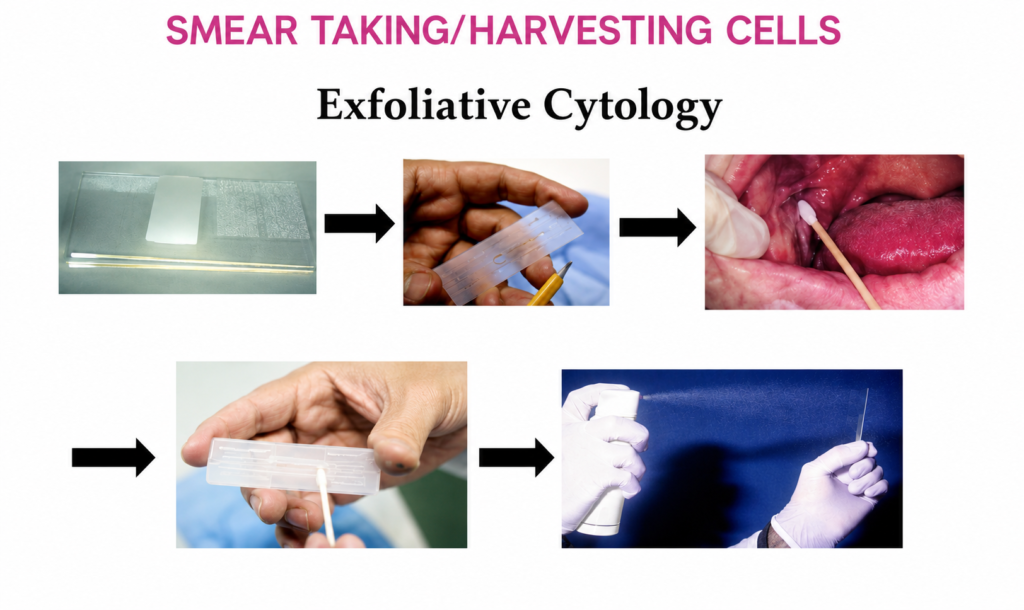
INTRODUCTION:- Screening of diseases gained significance in medicine at the end of the nineteenth century, when public health authorities emphasized the importance of screening methods for certain diseases. In 1941, George Papanicolaou demonstrated a test for the early detection of cervical cancer, contributing toward the creation of screening programs. Prevention and early diagnosis are major factors in reducing morbidity and mortality resulting from neoplasia. Screening of diseases involves a test or examination that can detect the existence of a particular disease in a high-risk population, asymptomatic or with minimum symptoms of the disease. CYTOLOGICAL SCREENING:- Screening of a particular disease requires a precise test, easy to do, at a low cost,and the capability of detecting the presence of a lesion. Cancer of the uterine cervix is an important cause of morbidity and mortality among women worldwide and a leading public health problem. It is the second most common cancer in women, but the most common in developing countries. Because of the phases that precede the lesion in the natural progress of invasive cervical cancer, and because they can be easily discovered and treated, the disease is well suited.to screening programs. The Papanicolaou test is an established method for examining the cells collected from the cervix to determine whether they show signs of pre-neoplastic differentiation. Cytologic screening programs have led to a large decline in cervical cancer incidence and mortality in developed countries. However, cervical cancer remains largely uncontrolled in high risk developing countries because of ineffective or no screening. Cervical cancer can be avoided when there is an early diagnosis of the precursor lesions, without local or systemic compromise. Among the methods available for early detection of cervical cancer, exfoliative cytology, or the Pap test, is recommended worldwide for mass screening, because the efficacy in the detection of premalignant lesions, associated with the social role of the method, permits minimization of costs with curative medicine. The basic integrated actions include: (1) care with collection, (2) processing of the smears, (3) screening and interpretation of the specimens, (4) follow-up of the patients, and (5) quality control. 1. Care with collection: The majority of false-negatives arise from problems with collection of specimens, and for this re 0ason this stage should be systemized and there should be training and recycling of the personnel responsible for taking the samples. The smears must be well identified, slim, uniform, and without contaminants, and contain samples from the transformation zone, where in the majority of cases the cervical cancer develops. There should be a minimum of blood, mucus, or other obscuring material such as lubricating gel. It is also important at this moment to adequately fix the material so as not to compromise subsequent stages. 2. Processing the specimens:- One of the characteristics of the Pap test is that it consists of various stages. Each stage should be monitored so as to minimize the possibility of error. The condition on arrival of the slides, and the number of slides per case, must be verified. Special care should be taken with the flow of the tests, with adequate numbering and balanced coloration with control of the number of cases colored in each set. The end product of this stage will be fundamental to a good result with the rest. 3. Screening and interpretation of the specimens:-The screening should be done in as little time as possible, depending on the basic requirements of each program, by trained and qualified personnel. Care should also be taken with excess workloads for cytopathologists and cytotechnicians, and also with refresher courses and recycling. The report on the tests should be systemized and use a unique nomenclature, of which all involved in the preparation and interpretation of the results should be fully aware. 4. Follow-up of patients:- Mere detection of the lesions will not determine the impact on the natural history of the disease. For this reason the treatment of lesions in a pre-invasive stage is fundamental.5. Quality control:- Quality is fundamental in gynecological cytopathology. One of the greatest problems in mass cytology is the false-negative cases. Cytopathology labs must have mechanisms for internal quality control with the objective of avoiding false-negative and false-positive tests. External quality control must be included in the design of the prevention program. THE ROLE OF LABORATORY IN SCREENING PROGRAMS:- The laboratory can make an important contribution to the structuring and organization of cervical screening programs based on the Papanicolaou test. The lab, when integrated into a screening program, should have among its objectives top quality production, training, and updating of personnel and the guarantee of a secure place of work, where risk factors are under control and the environment is protected. The system of internal monitoring of laboratory quality includes a set of actions, which should be developed and disseminated in a coordinated way, involving the various stages in the work process, from collecting a sample to issuing the report. The system aims to accompany and evaluate the cyto- and histopathologic diagnostic procedures in the laboratories, thus helping to determine areas where improvements can be planned and implemented, and also evaluate the impact of these actions and the incorporation of new practices.The majority of cervical cancer occurs in developing countries. The success of cervical cancer screening is shown by its ability to reduce the incidence of cervical cancer and the resulting mortality. The integration of procedures is essential for a successful screening program. Recently new technologies for alternative and complementary forms of screening such as liquid-based cytology and automated cytology have been proposed. A combination of methods has been proposed in an attempt to improve the sensibility of the Pap test. Among these, the association of cytology with the molecular test for HPV using hybrid capture has been highlighted. Automated cytology may be used for the purpose of reducing human errors caused by human fatigue, and to detect lesions with a lesser number of abnormal cells in the sample. HPV vaccine will be an additional tool in the strategies to reduce morbidity and mortality from cervical cancer and will be a component of a comprehensive strategy with the long-term goal of eliminating the disease. Cytologic screening can also be performed in selected high-risk