Calorimeter Principle,Components,Working & Applications:-
calorimeter:- A Colorimeter involves the measurement of Color and is the widely used method for finding the concentration of biochemical compounds. It Measures absorbance and wavelength between 400 to 700 nm (nanometer) i.e. from the visible spectrum of light of the electromagnetic spectrum.
Absorption of light :- Light falling on a colored solution is either absorbed or transmitted. A colored solution absorbs all the colors of white light and selectively transmits only
one color. This is its own color.
PRINCIPLE OF COLORIMETER:-
A colorimeter is based on the photometric technique which states that When a beam of incident light of intensity I0 passes through a solution, a part of the incident light is reflected (Ir), a part is absorbed (Ia) and rest of the light is transmitted (It)
Beer’s Law :-
This law states that the amount of light absorbed is directly proportional to the cocentration of the solute in the solution .
Lambert’s Law:-
The Lambert’s law states that the amount of light absorbed is directly proportional
to the length and thickness of the solution under analysis.
In simplified form,
The working principle of the colorimeter is based on Beer-Lambert’s law which states that the amount of
light absorbed by a color solution is directly proportional to the concentration of the solution and the length of a light path through the
solution.
A ∝ cl
Where,
A = Absorbance / Optical density ofsolution
c = Concentration of solution
l = Path length
A = ∈cl
∈ = Absorption coefficient
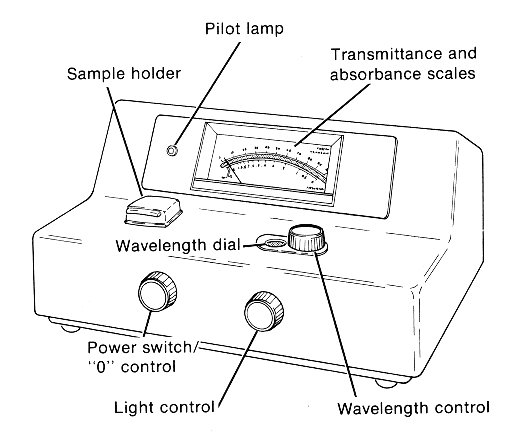
PARTS OF COLORIMETER:-
There are 5 essential parts in a calorimeter
Light Source – The most common source of light used in colorimeter is a tungsten filament.
Monochromator – To select the particular wavelength filter or monochromators are used to split the light from the light source.
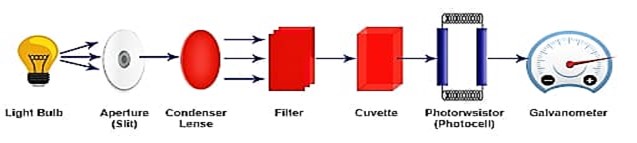
Sample holder –
Test tube or Cuvettes are used to hold the color solutions they are made
up of Glass at the visible wavelength.
Photo Detector System – when light falls on the detector system, an electric current is generated, this reflects the Galvanometer reading.
Measuring device – The current from the detector is fed to the measuring device, the Galvanometer, shows
the meter reading that is directly proportional to the intensity of light.
the formula used for determining the concentration of a substance in the test solution
A = ∈cl
For two solutions i.e. Test and standard,
∈ = Constant
l = Constant (using the same Cuvette or Standard cell)
AT = CT ….. (i)
AS = CS ….. (ii)
From (i) & (ii),
AT × CS = AS × CT
CT = (AT/AS) × CS
Where,
CT = Concentration of the Test solution
AT =Absorbance/ Optical density of the test solution
CS =Concentration of the standard
AS =Absorbance / Optical density of the standard solution